 These additional neutrons crash into other uranium 235 atoms, causing them to break apart and release additional neutrons, and on and on. 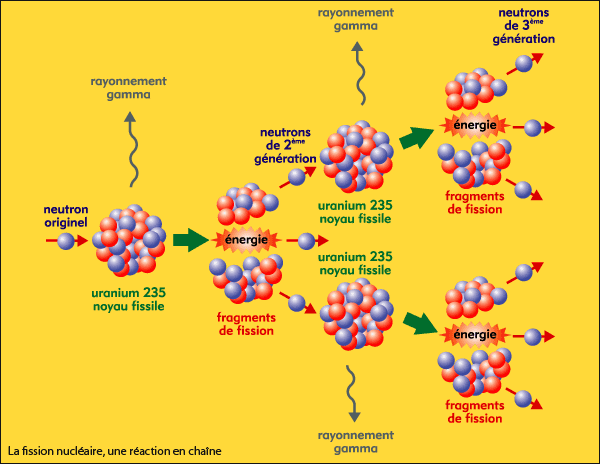
When hit, the nucleus absorbs the neutron, becomes unstable and decays, splits into two lighter atoms and throws out two or three new neutrons. Uranium 235 is more likely to undergo fission when hit by a slow-enough moving neutron. When it absorbs a neutron, it usually doesn’t break apart. Uranium 238 is the most common uranium isotope, and more stable. Uranium comes in two principle forms, or isotopes: uranium 235 and uranium 238. In a boiling water reactor, this heat becomes steam, which drives turbines to generate the electricity that is used for everything from charging smartphones to heating homes. As the nucleus splits, it releases energy, in the form of heat. Absorbing these excess neutrons sometimes causes the atoms to break apart. In reactors, fission occurs when uranium atoms are hit by slow-moving neutrons. Learn more about the use of Potassium Iodide (KI) in a radiation emergency.What is the difference between the nuclear material in a bomb, versus a reactor?Ī nuclear reactor works by using the energy that is released when the nucleus of a heavy atom splits. Once inside the body, radioactive iodine will be absorbed by the thyroid gland, potentially increasing the risk for thyroid cancer or other thyroid problems.Ī precaution against thyroid exposure in a radiation emergency is to take Potassium Iodide (KI). Consequently, people can receive internal exposure from drinking the milk or eating dairy products made from contaminated milk. In addition, if dairy animals consume grass contaminated with iodine, the radioactive iodine will be incorporated into their milk. If iodine were released into the atmosphere, people could ingest it in food products or water, or breathe it in. The thyroid gland uses iodine to produce thyroid hormones and cannot distinguish between radioactive iodine and stable (nonradioactive) iodine. Internal exposure can affect the thyroid gland, a small organ located in the neck near the Adam’s apple. Iodine and HealthĮxternal exposure to large amounts of iodine can cause burns to the eyes and skin. Learn about I-131 used in nuclear medicine. I-131 is often used to treat thyroid cancer. In medicine, I-131 is supplied in capsules or liquid form for patients to swallow. from nuclear weapons or reactor accidents can occur in particle form, which can be ingested in food or water. Iodine-131 in fallout falloutRadioactive material in the air from a nuclear explosion that will cool into dust-like particles and fall to the ground. Atmospheric testing in the 1950s and 60s released radioactive iodine to the atmosphere. Iodine-129 has dispersed around the world, and is now found at very low levels in the environment. Most I-129 in the environment came from nuclear weapons testing. Iodine-131’s short half-life of 8 days means that it will decay away completely in a matter of months. If released, I-129 will remain in the environment for millions of years. In soil, however, it combines easily with organic materials and moves more slowly through the environment. Radioactive iodine can disperse rapidly in air and water. Iodine readily combines with other elements and does not stay in its pure form once released into the environment. Iodine dissolves easily in water or alcohol. Iodine can change directly from a solid into a gas, skipping the liquid phase, in a process called sublimation. All 37 isotopes of iodine chemically interact with the environment in the same manner. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
